胆总管囊肿(choledochal cyst, CDC)又称先天性胆道扩张症,以胆总管部分或全部扩张为特征,可伴肝内胆管扩张,在亚洲人群中高发,发病率约1/13 000,且女性多于男性[1-2]。随着产前检查技术的发展,越来越多的CDC在胎儿期即被确诊[3]。由于产前识别的CDC患儿存在早发性肝纤维化风险,需及时进行临床评估与手术干预[4]。目前,产前识别CDC的手术决策仍存在争议。传统的“症状驱动”手术模式存在局限性,因临床症状的出现往往提示已发生一定程度的肝损害[4]。国内外关于CDC临床症状早期预警机制的研究尚显不足,本研究旨在明确产前识别CDC患儿出现临床症状的危险因素,为临床手术时机的选择提供客观参考依据。
资料与方法 一、一般资料本研究为回顾性研究,纳入2018年1月至2025年5月于福建省儿童医院接受手术治疗的CDC患儿。初始筛查产前识别的CDC患儿47例,排除分期手术1例、合并神经母细胞瘤1例、合并巨结肠类源病1例,最终纳入44例。患儿均行胆总管囊肿切除+肝总管空肠吻合术,随访3个月至6年。本研究经福建省儿童医院伦理委员会批准(2025ETKLRK10020),因回顾性研究豁免知情同意。
二、研究设计与观察指标根据术前是否出现呕吐、黄疸等临床症状,将44例患儿分为症状组(n=15)和非症状组(n=29);非症状组再根据手术年龄分为早期手术组(≤3个月,n=19)和晚期手术组(>3个月,n=10)。
观察指标:①基线资料(性别、诊断胎龄、手术年龄、手术时体重);②囊肿相关指标(Todani分型、术前囊肿最大长径/宽径、囊肿动态增长情况),其中囊肿的最大长径定义为囊肿最长的轴向距离,最大宽径定义为垂直于长度轴的最长距离;③术前肝功能指标(丙氨酸氨基转移酶ALT、天门冬氨酸氨基转移酶AST、GGT、DBIL);④手术及术后相关指标(手术时间、术后住院时间、术后肝活检病理分级(采用Batts-Ludwig分级系统[5])、术后并发症(吻合口瘘、吻合口狭窄、胆管炎、肠梗阻等)。
三、统计学处理采用SPSS 23.0进行统计学分析。正态分布的计量资料以x±s表示,组间比较采用独立样本t检验;非正态分布的计量资料以M(Q1, Q3)表示,组间比较采用Mann-Whitney U检验;计数资料以频数(例)描述,组间比较采用χ2检验或Fisher精确概率法。采用Pearson相关性分析探讨囊肿大小与DBIL的关系,通过ROC曲线评估囊肿大小对临床症状的预测价值。P<0.05为差异具有统计学意义。
结果两组患儿在性别、手术年龄、手术时体重、ALT值、AST值、手术时间、术后并发症比较,差异均无统计学意义(P>0.05)。两组患儿诊断胎龄、Todani Ⅳ型囊肿比率、手术前囊肿最大长径和宽径、GGT值、DBIL值、术后住院时间比较,差异均有统计学意义(P<0.05)。见表 1。
| 表 1 两组胆总管囊肿患儿基线资料与手术并发症比较 Table 1 Comparison of baseline profiles and surgical complications between two groups of children with choledochal cysts |
|
|
非症状组病例中,早期手术组(≤3个月)与晚期手术组(>3个月)比较,在囊肿大小、术前肝功能指标、肝纤维化程度、手术时间、术后住院时间、术后并发症发生率均无明显差异(P>0.05)。见表 2。
| 表 2 非症状组患儿不同手术时机的基线资料与手术并发症比较 Table 2 Comparison of baseline profiles and surgical complications in non-symptomatic group at different surgical timings |
|
|
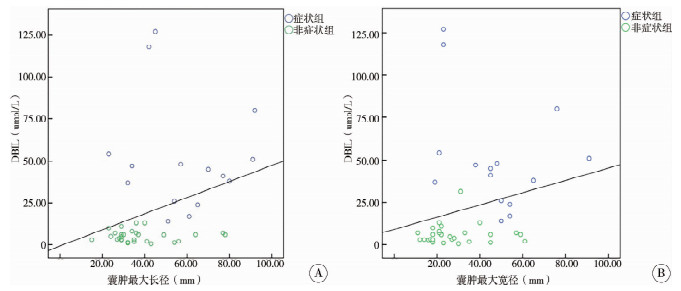
相关性分析显示,症状组和非症状组患儿术前囊肿最大长径与DBIL值呈正相关(r=0.364,P=0.015),术前囊肿最大宽径与DBIL值亦呈正相关(r=0.327,P=0.030)。见图 1。
|
图 1 症状组与非症状组患儿术前囊肿大小与DBIL相关性分析 Fig.1 Correlation analysis between preoperative cyst size and DBIL in symptomatic and asymptomatic groups 注 A:囊肿最大长径;B:囊肿最大宽径 |
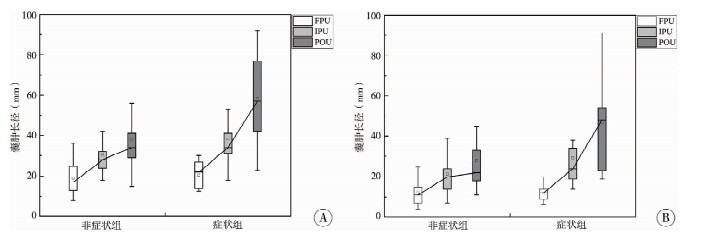
动态监测两组的囊肿最大长径从产前首次彩超[22(14, 27)比17(13, 25)]、生后首次彩超[34(31, 41)比28(23, 33)]至术前彩超[57(42, 77)比34(29, 42)],囊肿最大宽径从产前首次彩超[12(9, 14)比11(6, 15)]、生后首次彩超[24(19, 34)比20(14, 24)]至术前彩超[48(23, 54)比22(18, 34)],囊肿长径、宽径均呈逐渐增长趋势,且症状组的囊肿长径、宽径增长速度均快于非症状组。见图 2。
|
图 2 症状组与非症状组患儿囊肿长径和宽径增长趋势。 Fig.2 Growth trends of cyst length and width in symptomatic and asymptomatic groups 注 A:囊肿长径;B:囊肿宽径;FPU:首次发现产前彩超;IPU:生后首次彩超;POU:术前彩超 |
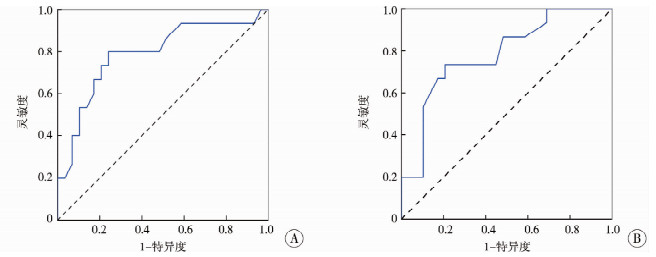
ROC曲线分析显示,术前囊肿最大长径预测临床症状的曲线下面积为0.789(95%CI:0.637~0.940),最佳截断值为44 mm,灵敏度73%,特异度79%;术前囊肿最大宽径的曲线下面积为0.783(95%CI:0.639~0.926),最佳截断值为36 mm,灵敏度73%,特异度81%(图 3)。
|
图 3 术前囊肿最大长径与宽径预测临床症状的ROC曲线图。 Fig.3 ROC analysis results of preoperative maximum length and width of cysts in predicting clinical symptoms 注 A:囊肿长径;B:囊肿宽径 |
手术切除是CDC唯一根治手段[6]。若延误治疗,可能引发胆管炎、囊肿穿孔、胆汁淤积性肝硬化等严重并发症,因此,早期识别临床症状危险因素并及时干预,对减轻肝纤维化、改善患儿预后至关重要[7]。成人及大龄儿童CDC的典型症状为腹痛、腹部包块、黄疸,但约1/3患者长期无明显症状[8];而产前识别的CDC患儿以梗阻性黄疸为主要表现,本研究中症状组患儿胆红素水平显著高于非症状组,这与胆道梗阻导致的胆汁淤积密切相关,胆汁淤积是CDC肝纤维化的核心病理过程,提示产前识别的CDC患儿生后可能存在持续肝损伤风险[4]。由于CDC患儿生后症状出现时间存在个体差异,部分患儿生后即刻发病,部分则延迟至数月后,给临床手术决策带来挑战[9-10]。
目前,无症状CDC患儿的手术时机尚存争议:日本指南推荐3~6月龄手术国内指南建议3个月内行根治术,既往部分研究因顾虑新生儿期麻醉及手术风险,主张推迟至3~6月龄手术[7, 10-12]。但随着腹腔镜与麻醉技术的进步,新生儿期手术并未增加术后并发症[13-14]。本研究中,非症状组早期手术与晚期手术的短期结局(手术时间、住院时间、并发症发生率)无显著差异,且晚期手术组未观察到肝损害加重,但早期手术的长期获益仍需更长时间随访验证。需注意的是,部分无症状患儿在等待手术期间可能出现临床症状,因此明确症状相关危险因素尤为重要[15]。本研究发现,症状组产前诊断胎龄早于非症状组,与杨丹[16]的研究结论一致,推测可能与胆汁淤积程度及囊肿发育时间相关。症状组中Todani Ⅳ型囊肿占比更高,由于Ⅳ型CDC存在肝内外胆管多发扩张,常合并肝内胆管狭窄及胰胆管合流异常,更易引发梗阻性黄疸[17]。GGT作为胆管上皮细胞特异性酶,与胆道梗阻密切相关,是CDC临床症状的重要预测指标[15],李思源等[18]曾报道GGT≥150.85 IU/L是CDC穿孔的独立危险因素,联合囊肿体积增长速率可提升预测效能。
囊肿大小是预测CDC症状的关键指标,但不同研究的阈值存在差异:Pan等[19]发现症状组囊肿平均大小(44.33 mm) 显著大于无症状组(30.34 mm);Cochran等[20]提出生后首次超声囊肿≥4.5 cm对1个月内症状的预测价值最高(曲线下面积0.744);另有研究认为生后囊肿直径>30 mm是梗阻性黄疸的预测因子[9]。本研究通过ROC曲线分析确定囊肿长径>44 mm、宽径>36 mm为临床症状的预测阈值,与上述研究存在差异,可能与样本特征、检测时机等因素相关。此外,本研究发现囊肿大小与DBIL虽呈正相关,但相关性较弱,提示胆道梗阻不仅与囊肿扩张程度有关,还可能受蛋白栓、胰胆管合流异常等其他病理因素影响,单一时间点的囊肿大小预测存在局限性[21]。动态监测显示,症状组囊肿增长速度快于非症状组,提示囊肿快速增大可能反映胆管内压力升高、胆汁排出受阻,更易出现梗阻相关症状。李思源等[18]也发现囊肿体积每周增长≥1.00 cm3是穿孔的独立危险因素,因此,“动态监测- 阈值量化”的个体化方案可能更适用于CDC临床症状的预测。
综上,产前识别的CDC患儿建议在无症状期接受手术治疗;产前诊断胎龄早、Todani Ⅳ型囊肿是生后出现临床症状的危险因素;囊肿快速增长及大小阈值(长径>44 mm、宽径>36 mm)可作为手术时机选择的重要参考。本研究存在一定局限性:①为单中心回顾性研究,样本量相对有限;②随访时间较短,对手术时机的长期影响(如远期胆管并发症、生长发育、麻醉对神经发育的潜在影响)尚未明确。未来需开展多中心、大样本量前瞻性研究,进一步验证研究结论。
利益冲突 所有作者声明不存在利益冲突
作者贡献声明 郑伟军、朱岚欣、刘明坤、陈飞负责文献检索,郑伟军、吴典明、刘明坤负责论文调查设计,郑伟军、朱岚欣负责故据收集与分析,郑伟军、方一凡、刘明坤负责论文结果撰写,郑伟军、方一凡、吴典明负责论文讨论分析,并对文章知识性内容进行审阅
| [1] |
Takahashi T, Shimotakahara A, Takahashi T, et al. Choledochal cyst associated with an accessory hepatic duct identified by intra-operative endoscopy: case report and literature review[J]. Pediatr Surg Int, 2008, 24(9): 1079-1082. DOI:10.1007/s00383-008-2213-x |
| [2] |
Yin T, Chen SY, Zhou RJ, et al. Relationships of serum MMP-7 and clinical characteristics in choledochal cyst children[J]. BMC Surg, 2024, 24(1): 195. DOI:10.1186/s12893-024-02488-y |
| [3] |
Kowalski A, Kowalewski G, Kaliciński P, et al. Choledochal cyst excision in infants-a retrospective study[J]. Children (Basel), 2023, 10(2): 373. DOI:10.3390/children10020373 |
| [4] |
Zhuang XJ, He YB, Wu DM, et al. Risk factors analysis for significant liver fibrosis of prenatally diagnosed choledochal cysts: a retrospective case-control study[J]. Front Pediatr, 2025, 13: 1595238. DOI:10.3389/fped.2025.1595238 |
| [5] |
Goodman ZD. Grading and staging systems for inflammation and fibrosis in chronic liver diseases[J]. J Hepatol, 2007, 47(4): 598-607. DOI:10.1016/j.jhep.2007.07.006 |
| [6] |
Weng R, Hu W, Cai S, et al. Prenatal diagnosis and prognosis assessment of congenital choledochal cyst in 21 cases[J]. J Obstet Gynaecol, 2016, 36(3): 324-327. DOI:10.3109/01443615.2015.1050648 |
| [7] |
黄挺, 刁美. 产前诊断胆总管囊肿的临床诊疗进展[J]. 中国医学前沿杂志(电子版), 2024, 16(3): 72-76. Huang T, Diao M. Recent advances in clinical diagnosis and treatment of choledochal cyst diagnosed prenatally[J]. Chinese Journal of the Frontiers of Medical Science (Electronic Version), 2024, 16(3): 72-76. DOI:10.12037/YXQY.2024.03-13 |
| [8] |
Nakagawa Y, Uchida H, Shirota C, et al. Characteristics and outcomes of minimally invasive surgery for congenital biliary dilatation in children aged < 6 years: comparison between children and adults[J]. J Hepatobiliary Pancreat Sci, 2024, 31(12): 876-885. DOI:10.1002/jhbp.12069 |
| [9] |
Shirota C, Hinoki A, Tainaka T, et al. Surgical strategies for neonates with prenatally diagnosed congenital biliary dilatation[J]. J Pediatr Surg, 2024, 59(3): 385-388. DOI:10.1016/j.jpedsurg.2023.10.045 |
| [10] |
Tanaka H, Sasaki H, Wada M, et al. Postnatal management of prenatally diagnosed biliary cystic malformation[J]. J Pediatr Surg, 2015, 50(4): 507-510. DOI:10.1016/j.jpedsurg.2014.08.002 |
| [11] |
Ishibashi H, Shimada M, Kamisawa T, et al. Japanese clinical practice guidelines for congenital biliary dilatation[J]. J Hepatobiliary Pancreat Sci, 2017, 24(1): 1-16. DOI:10.1002/jhbp.415 |
| [12] |
中华医学会小儿外科分会腔镜外科学组. 腹腔镜胆总管囊肿手术操作指南(2017版)[J]. 中华小儿外科杂志, 2017, 38(7): 485-494. Section of Laparoscopic & Endoscopic Surgery, Branch of Pediatric Surgery, Chinese Medical Association. Guideline for laparoscopic hepatojejunostomy for choledochal cyst in children (2017)[J]. Chin J Pediatr Surg, 2017, 38(7): 485-494. DOI:10.3760/cma.j.issn.0253-3006.2017.07.002 |
| [13] |
Rong LY, Li YB, Tang JF, et al. Robotic-assisted choledochal cyst excision with Roux-en-Y hepaticojejunostomy in children: does age matter?[J]. Surg Endosc, 2023, 37(1): 274-281. DOI:10.1007/s00464-022-09496-0 |
| [14] |
Jin Y, Chen QJ, Zhang YB, et al. Robot-assisted resection of choledochal cysts in children weighing less than 6 kg[J]. Br J Surg, 2023, 110(2): 267-268. DOI:10.1093/bjs/znac361 |
| [15] |
Yang D, Li L, Diao M, et al. Risk factors analysis for clinical symptoms of prenatally diagnosed choledochal cysts: a retrospective study[J]. BMC Surg, 2023, 23(1): 217. DOI:10.1186/s12893-023-02115-2 |
| [16] |
杨丹. 产前诊断的儿童胆总管囊肿临床特征分析[D]. 北京: 北京协和医学院, 2024. DOI: 10.27648/d.cnki.gzxhu.2024.000363. Yang D. Analysis of clinical features of prenatally diagnosed pediatric choledochal cyst[D]. Beijing: Peking Union Medical College, 2024. DOI: 10.27648/d.cnki.gzxhu.2024.000363. |
| [17] |
Jang SM, Lee BS, Kim KK, et al. Clinical comparison of choledochal cysts between children and adults[J]. Korean J Hepatobiliary Pancreat Surg, 2011, 15(3): 157-163. DOI:10.14701/kjhbps.2011.15.3.157 |
| [18] |
李思源, 刁美, 李龙. 超声联合谷氨酰转肽酶检测对产前诊断的胆总管囊肿穿孔风险的预测价值研究[J]. 临床小儿外科杂志, 2024, 23(12): 1149-1154. Li SY, Diao M, Li L. Application value of natal ultrasonographic combined with gamma-glutamyl transpeptidase testing in assessing the risk of choledochal cyst perforation[J]. J Clin Ped Sur, 2024, 23(12): 1149-1154. DOI:10.3760/cma.j.cn101785-202405033-008 |
| [19] |
Pan SW, Li W, Chen H, et al. The timing of minimally invasive surgery for prenatally diagnosed choledochal cysts[J]. BMC Pediatr, 2024, 24(1): 250. DOI:10.1186/s12887-024-04734-x |
| [20] |
Cochran ED, Lazow SP, Kim AG, et al. The in-utero diagnosis of choledochal cyst: can postnatal imaging predict benefit from early surgical intervention?[J]. J Matern Fetal Neonatal Med, 2022, 35(6): 1070-1074. DOI:10.1080/14767058.2020.1742320 |
| [21] |
Gang SJ, Kim PH, Kwon H, et al. Risk factors for postoperative stone formation in pediatric choledochal cysts: a study of 457 cases[J]. Sci Rep, 2025, 15(1): 9235. DOI:10.1038/s41598-025-94308-3 |
 2026, Vol. 25
2026, Vol. 25


